It's a New Day in Public Health.
The Florida Department of Health works to protect, promote, and improve the health of all people in Florida through integrated state, county, and community efforts.
HIV Data Center
HIV/AIDS
no content in this box
HIV/AIDS Surveillance Program
The goal of the HIV/AIDS Surveillance Program is to collect complete and accurate HIV data to analyze trends. HIV staff and partners use these data and trends to plan, carry out, and assess HIV programs statewide.
People with HIV (PWH) may be tested often to monitor treatment needs. This can lead to single cases being reported multiple times. Surveillance staff regularly check data to correct for this, both within Florida and within the CDC’s national database. Duplicate cases within the state are merged.
PWH may move out of state or out of the country one or several times during their lifetime. Cases identified as being reported first in another state are re-assigned as that state’s case. Cases for people who have resided in multiple states are identified either by comments on the Case Report Form or through the CDC's Routine Interstate Duplicate Review (RIDR) process. These deduplication activities are part of the ongoing surveillance process that occurs in every state.
The entire surveillance HIV/AIDS data set is reviewed in the deduplication process. That means that changes made affect both recently reported case counts and those from prior years. Each year, the HIV data for the previous calendar year and all previous years are finalized and frozen for reporting purposes on June 30. The frozen data are used in all data reports until the following June 30, when the continuously deduplicated HIV/AIDS data set will be finalized and frozen again.
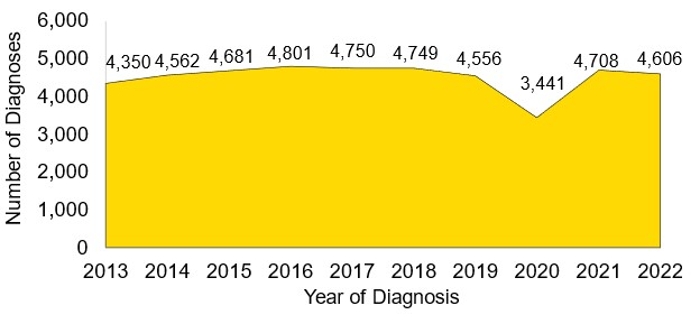
Figure 1 (below) depicts a summary of newly diagnosed cases of HIV from the last ten years as of June 30, 2023. Summary health data are stored in the Florida Community Health Assessment Resource Tool Set (Florida CHARTS), Florida Health’s database for tracking the health status of the state. Visit Florida CHARTS to learn more about statistics for HIV, AIDS, and HIV-related deaths.
Data for 2020 and 2021 should be interpreted with caution due to the impact of COVID-19 on HIV testing, care-related services, and case surveillance activities.
Figure 1
Diagnoses of HIV, 2013–2022, Florida
Diagnosis or Report?
Florida tracks HIV/AIDS cases by the year they were diagnosed and by the year they were reported.
- Year of diagnosis is the year a person is first diagnosed with HIV/AIDS.
- Year of report is the year a person's case is first reported to Florida Health and entered into the enhanced HIV/AIDS Reporting System.
The year of diagnosis and the year of report may be different for each case because there are specific requirements set by the CDC for when an HIV/AIDS case is considered reportable, even though the person has been diagnosed. There are many criteria that determine whether or not a case is reportable.
The following are examples of how HIV cases are diagnosed and reported:
- Example 1:
- Example 2:
HIV Care Data
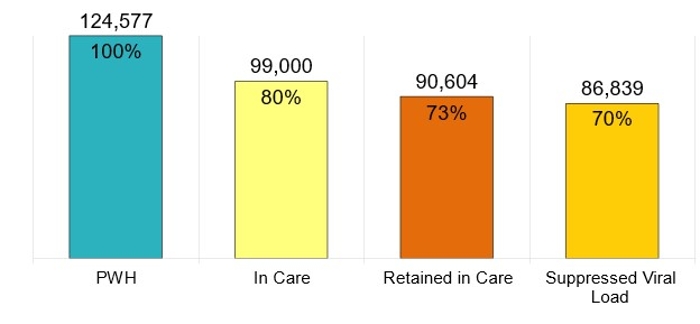
While it is important to understand the number of new HIV cases in Florida, it is also important to be aware of the overall number of people living with HIV in the state. We track this data because it informs decisions related to resource allocation to ensure all PWH in Florida have access to the care they need. Figure 2 presents the levels of HIV care of the people living with HIV in Florida. This model is updated annually.
Figure 2
PWH Living in Florida
Along the HIV Care Continuum, Year-End 2022
Care Continuum Definitions
- PWH: The number of persons with an HIV diagnosis living in Florida at the end of 2022
- In Care: PWH with at least one documented viral load or CD4 lab, medical visit, or prescription from 1/1/2022 to 3/31/2023
- Retained in Care: PWH with two or more documented viral load or CD4 labs, medical visits, or prescriptions at least three months apart from 1/1/2022 to 6/30/2023
- Suppressed Viral Load: PWH with a suppressed viral load (<200 copies/mL) on the last viral load test from 1/1/2022 to 3/31/2023



Connect with DOH